GE HealthCare’s recently release financials for full year 2025 reveal that the company’s imaging vertical contributed…
Continue ReadingTag: FDA
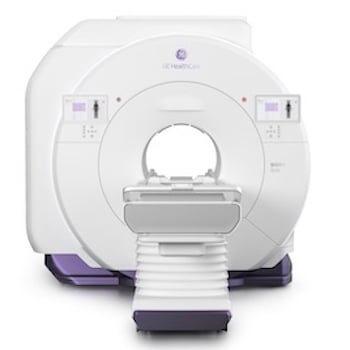
GE HealthCare’s SIGNA MRI Portfolio Granted FDA Clearance
Feb. 19, 2026 — GE HealthCare recently announced 510(k) clearance of three new magnetic resonance (MR) innovations…
Continue Reading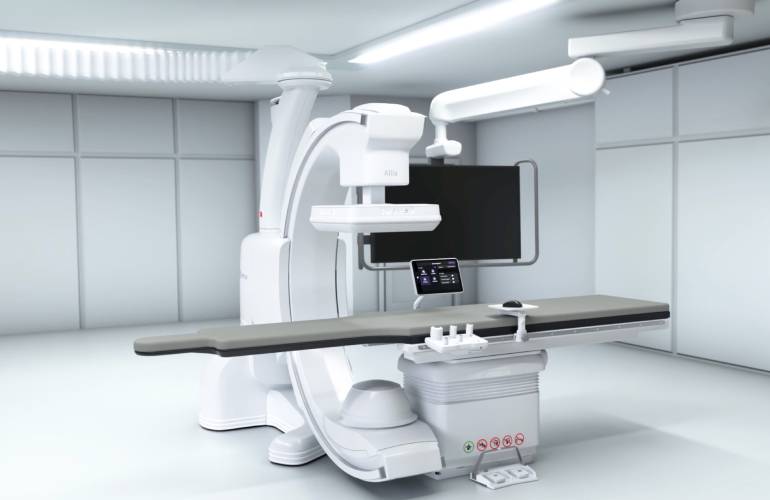
GE HealthCare wins FDA, CE mark approval for Allia Moveo
An animated rendering of the Allia Moveo platform. [Image from GE HealthCare] GE HealthCare (Nasdaq: GEHC)…
Continue Reading
GE HealthCare wins FDA clearance for Allia Moveo interventional imaging system
GE HealthCare has received U.S. Food and Drug Administration 510(k) clearance and CE Marking for Allia…
Continue Reading
GE Healthcare’s Allia Moveo Platform Receives FDA Clearance, CE Mark
Feb. 2, 2026 — GE HealthCare has announced that Allia Moveo has received U.S. Food and Drug…
Continue Reading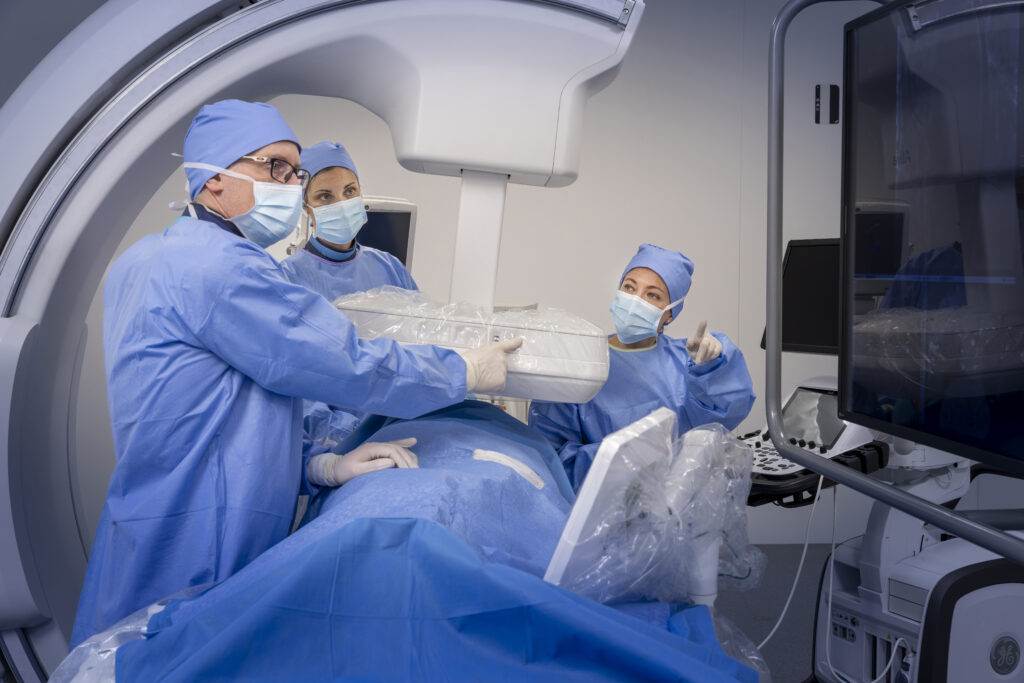
GE HealthCare’s Allia Moveo gains FDA and CE mark approvals
GE HealthCare’s Allia Moveo system GE HealthCare has announced that its Allia Moveo image-guidance platform has…
Continue Reading
