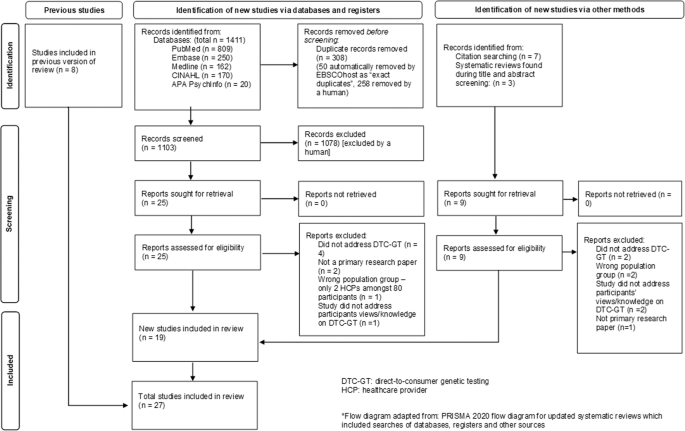
A total of 1411 records were retrieved from the databases. Of these, 308 duplicates were removed, resulting in 1103 records for title and abstract screening. Following exclusion of ineligible records, 25 full texts were reviewed, of which 17 papers met the eligibility criteria (details of 8 excluded full texts are provided in Appendix A). Two additional papers from other sources were found to fulfil the criteria for inclusion [21, 22]. Therefore, 19 new papers were identified in this review. Including the eight reports identified in the 2012 review [7] this updated systematic review identified 27 reports. Further details on study screening processes can be found in the flow diagram Fig. 1. There were two instances where two reports related to the same study (two newly identified records [22, 23], and two identified by Goldsmith et al. [7]. Therefore, this review identified a total of 25 studies that address the knowledge and views of HCPs on DTC-GT for further evaluation. The appraisal of the new studies informed by PRISMA 2020 is included in Fig. 2.
The PRISMA diagram* depicts our study selection process which includes the previous studies identified and the identification of new studies via databases, registers and other methods [8].
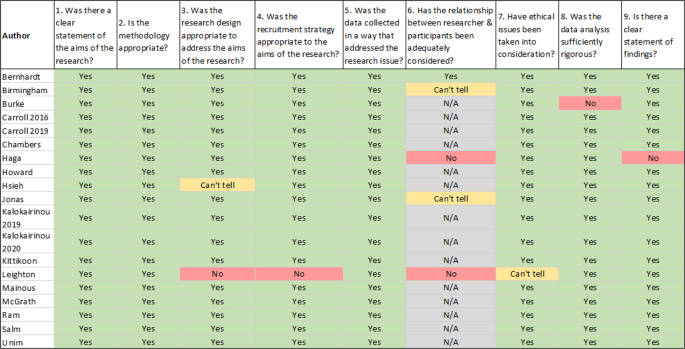
Evaluation of each included study based on the nine criteria included in the CASP Qualitative studies tool [12].
Study characteristics
The main characteristics of the 18 new studies, covered in 19 reports, are included in Table 2. Of these, fourteen studies were conducted in North America, either in the USA, Canada, or both [21, 24,25,26,27,28,29,30,31,32,33,34,35,36]. Two studies were conducted in Europe and involved HCPs from several European countries [2, 22, 23]. One study was conducted in New Zealand [37], and one in Thailand, the only one conducted in a low-to-middle income country [38].
Sixteen studies used survey methods, of which eleven were online [2, 24, 26, 29,30,31,32,33,34,35,36], three by mail [21, 28, 37], one on paper [38], and one used both mail and on paper [25]. Two studies involved focus groups [25, 27], one with a pre- and post-focus group survey [25], and one used semi-structured interviews [22, 23].
The response rates in the survey-based studies varied considerably. Eight studies had less than a 20% response rate [26, 29,30,31,32, 34,35,36], six studies had between 21 and 50% response rate [2, 21, 24, 28, 33, 37], and one study had a response rate of 60% [38]. Similarly, participant numbers varied across the survey-based studies; six had less than 200 participants [2, 26, 29, 32, 35, 37], six studies had between 201 and 500 participants [21, 28, 30, 34, 36, 38], one study had 502 participants [24], and two studies had more than 1000 participants [31, 33]. The two focus group studies had 24 and 51 participants, and the semi-structured interview study had 15 participants [22, 23, 25, 27].
The types of participants involved in these studies were diverse. Participant groups from each study were divided into one or more of the following categories: (1) medical specialists, (2) GCs, (3) clinical geneticists (CGs), and (4) primary care physicians (PCPs), including a small number of registered nurses, nurse practitioners and physician assistants. Twelve studies involved PCPs [21, 24, 25, 27,28,29, 31, 33,34,35, 37, 38]. Four studies had medical specialists in their participant groups: one study involved psychiatrists and neurologists [36], one study involved urologists [25], and two involved a small number of other medical specialists [21, 34]. Five studies involved GCs [26, 30, 32, 34, 35]. Two studies involved only CGs as participants [22, 23, 35] and one study had CGs represent 1.5% of their participant group [34]. The two studies that only involved CGs were both conducted in Europe.
Regarding risk of bias, the included reports were of good quality (see Fig. 2). Six of the 19 papers had at least one issue, the most common being that the relationship between the researcher and the participants was not adequately considered, followed by the research design not being appropriate to address the aims of the research.
Main findings
The main findings identified in each of the new papers are included in Appendix B. These findings have been divided into the same four themes identified by Goldsmith et al.
DTC-GT awareness, knowledge, and experiences
Awareness
Awareness of DTC-GT was understandably higher amongst genetic specialists (GCs and CGs) compared with other HCPs. Over 95% of GCs had heard of DTC-GT before taking part in a study conducted in the US [32]. Eighty-six per cent of CGs were aware of DTC-GT services. Sixty-four per cent of those aware of these services could name at least one company that offered them [2]. Forty-eight per cent of general practitioners in New Zealand had heard about DTC-GT [37]. Fifteen per cent of internal medicine physicians in Thailand were aware of DTC-GT [38]. Related to specific DTC tests, 50% of urologists and PCPs had not heard of or read about DTC-GT for prostate cancer [25] and only 23% (n = 7) of the participants in a Canadian study were aware that patients interested in DTC-GT were able to receive counselling and other services in their healthcare facilities [35].
Knowledge
Knowledge and confidence about DTC-GT was higher amongst HCPs specialising in genetics when compared with other HCPs. This was, however, not consistent throughout the studies. Seventy-four percent of a group of PCPs versus 83% of genetic specialists were able to correctly interpret DTC-GT results [34]. Fifty-eight per cent of these PCPs versus 95% of genetic specialists agreed or strongly agreed that that they were able to understand and discuss genetics related information, and 46% versus 86% were somewhat prepared or well prepared to discuss DTC-GT results with patients [34]. The included studies identified considerable variation in knowledge and confidence with DTC-GT across and between HCPs, with CGs and PCPs reporting both high and low levels of knowledge and confidence in their ability to understand, interpret, and communicate results with patients surrounding DTC-GTs [21, 22, 24, 25, 27, 28, 32, 38]. More specifically, while one study identified that only 16% of PCPs felt that DTC-GT results were too difficult to understand [38], another study with reported that 96% of participating urologists and PCPs did not feel confident and 79% did not feel prepared to answer patients’ questions about DTC-GT [25]. One study identified that HCPs who had personal experience with DTC-GT had improved knowledge of DTC-GT and were more likely to recommend DTC-GT to patients [29].
Experience
Genetic specialists had more DTC-GT experience than other HCPs, with 58% of genetic specialists versus 17% of PCPs had experience with patients bringing them personal DTC-GT results [34]. Three studies involving GCs looked at experience with patients inquiring about DTC-GT, with 40% to 94% of GCs having encountered questions related to DTC-GT results review [26, 30, 32]. Conversely, an earlier study reported that only about one quarter of GCs had previous experience with a patient asking about DTC-GT results [32]. Overall, PCPs and other non-genetic specialists (e.g. neurologists) had fewer experiences with patients presenting with DTC-GT results, with a range of 8% to 15% of HCPs reporting experience with patients inquiring about DTC-GT results [22, 24, 25, 36]. Only 5% of PCPs had five or more patients share DTC-GT health risk results with them in the past year, while 30% had five or less patients share these results with them, and 65% did not have any [31]. For family physicians, 71% had never had patients ask them about DTC-GT, while 28% had patients ‘rarely’ ask them about DTC-GT [33]. One study mentioned healthcare providers’ own experiences with DTC-GT, showing that 16% of GCs had undergone DTC-GT themselves and 9% had experience with it on behalf of their relatives [30].
Direct-to-consumer genetic testing beliefs and opinions
Clinical utility and validity of DTC-GT
Compared to CGs, more PCPs and medical specialists believed that DTC-GT was useful in the clinical setting. Most CGs emphasised that the clinical utility and validity of genetic tests should be a fundamental condition when made available to the public. However, many CGs felt that DTC-GT results were inconsequential for their patients and that DTC companies ignored consumers’ family history [23]. Further, CGs were more reluctant to trust DTC-GT results than other HCPs [22, 34, 37].
Alternatively, the perspectives of clinical utility and validity from PCPs and non-genetic specialists is mixed. Studies identified that most PCPs and urologists agreed that DTC-GT results could assist in decision making on the initiation and frequency of prostate cancer screening (86% and 76%, respectively) [25], as well as that 40% of PCPs felt that DTC-GT results would help in managing a patient [24]. Alternatively, results from another study identified that 39% of PCPs felt that DTC-GT had no clinical utility and 20% would not change patient management based on DTC-GT results [38]. Furthermore, in one study, 67% of PCPs felt that one of the barriers to discussing DTC-GT results was its lack of current relevance in clinical decision making [21].
Direct-to-consumer access to genetic testing
CGs have been consistent with regards to restricting access to certain genetic tests outside of a clinical setting. Howard and Borry reported that 90% of CGs either somewhat disagreed or strongly disagreed that testing for disorders that were preventative or treatable should be accessible directly to the public, with similar opinions surrounding testing for conditions or traits with no or somewhat mild health consequences [2]. Another study involving GCs identified only a small percentage of GCs would find DTC-GT acceptable for testing adult-onset conditions (7%) and cancer (6%). Additionally, most GCs (96%) stated that some forms of DTC-GT are acceptable [30].
Despite ease of access and availability, most CGs felt that it was a necessity for HCPs to be involved in the health-related genetic testing process [22], that DTC genetic tests should hold up to the same quality as tests offered by the healthcare system, and that medical supervision and genetic counselling should be compulsory in some form [23]. CGs were split as to who should supervise this process as some felt that it did not have to be a medical doctor but instead could be another qualified medical professional or a nurse. CGs were also split on whether involving HCPs employed directly by DTC-GT companies would be appropriate or would create a conflict of interest [23].
Beliefs that DTC-GT had positive and negative effects on consumers
HCPs expressed beliefs about positive effects of DTC-GT on consumers, with over two-thirds of PCPs indicating that there were multiple benefits to DTC testing (e.g. motivating a healthy lifestyle, detecting adult-onset diseases earlier, and assisting patients in becoming more proactive in their own health) [29]. In another study, general practitioners (GPs) believed that the following were perceived benefits to DTC-GT (convenience, promotion of preventative medicine, confidentiality of results); however, participants tended to disagree that DTC-GT provided a useful service [37]. Finally, 39% of GCs felt that DTC-GT had value, with some of the reasons being consumer access to genetic testing, increasing genetics knowledge, and providing valuable data for research use [30].
Several studies reported various concerns that HCPs had regarding the negative effects of DTC-GTs on consumers. Compared with PCPs, GCs seemed to have more concern about the harm that DTC-GT may have on consumers, with 91% of GCs disagreeing with the statement that DTC-GT caused no harm [30]. While 58% of PCPs (family physicians) felt that DTC-GT is more likely to cause harm than to provide a benefit to patients, 10% felt that it was likely to help patients and 32% felt that it was unlikely to make any difference [33]. Additionally, CGs in Europe felt that advertising by DTC companies was misleading and ‘even manipulative’, suggesting information available on company websites was often deficient [23].
DTC-GT and healthcare professional involvement
Both genetic specialists and PCPs were uncomfortable or lacked some confidence in providing genetic counselling pertaining to DTC-GT to patients. Although 91% of GCs reporting that DTC-GTs could be improved by genetic counselling, only 31% felt that they would be comfortable in providing counselling to patients with DTC-GT results [30]. Of the GCs that had experience in encountering patients with DTC-GT, approximately 33% felt negatively about these encounters, most commonly citing that DTC-GT was not the best use of clinical time and the GCs did not know how to interpret such results [30]. Just over half of the GCs felt some degree of negativity towards DTC-GT, while only 7% felt some degree of positivity about DTC-GT [30].
GCs and PCPs felt it was their responsibility to counsel patients on the benefits and risks of genetic testing, despite difficulties and frustrations with their DTC-GT involvement [2, 27]. Similarly, 62% of GPs felt that they should be involved in the DTC-GT process even though participants in this study tended to agree that DTC-GT ‘negatively impacts the [doctor]–patient relationship’ [37]. GPs felt that the following were perceived barriers for them to provide genetic counselling (expressed in the percentage who agreed with each issue): time (81%), experience (91%) and knowledge (89%) [37].
DTC-GT in the future
Overall, a minority of PCPs (49%) felt that DTC-GT felt would be commonplace in the next 5 years [24], with 58% of New Zealand unsure if DTC-GT would be an area of growth [37]. Despite this uncertainty, another study identified that 58% of PCPs (family physicians) were interested in new technological advances like DTC-GT [28].
Downstream costs and referrals due to DTC-GT
Several studies suggest how DTC-GT leads to an increased number of referrals and increased downstream costs (e.g. disease screening) by means of more follow-up visits or investigations, confirmatory genetic testing or other unnecessary procedures. In a group of PCPs and specialist physicians who had experience with patients sharing DTC-GT results, 40% made at least one referral to a specialist, with 78% of referrals to a CG or GC [31]. Additionally, the results of one study identified that 60% of internal medicine physicians felt that DTC-GT could assist in clinical decision making and that two-thirds of these physicians felt that DTC-GT could lead to earlier or more frequent disease screening in an at-risk population [38]. Further, most GCs would order confirmatory genetic testing for a patient presenting to their clinic with an Ashkenazi Jewish founder mutation in Breast Cancer gene (BRCA)1/2, irrespective of the patient’s pedigree [26]. GPs tended to agree that DTC-GT results encouraged patients to get unnecessary procedures and genetic tests while suggesting that DTC-GT puts pressure on healthcare resources that could be used for something else [37]. GPs (81%) felt that time was an anticipated barrier for them to provide genetic counselling [37]. Similarly, PCPs in one study felt that the lack of clinical time and the lack of reimbursement were some of the barriers when discussing DTC-GT results [21].
Genetic counsellors’ views of their roles in DTC-GT
GCs gave their opinions on the following possible challenges related to providing genetic counselling for DTC results in a clinical setting (percentage who found it easy compared with neutral or difficult): 1) explaining that clinical grade genetic testing was required to confirm results (86%), 2) explaining the differences between clinical and SNP-based testing (61%) and 3) managing patient anxieties and expectations (39%) [26]. In another study, 31% of GCs felt comfortable providing counselling to patients who had undergone DTC-GT, while 26% were neutral and 42% did not agree with this statement [30].
When considering how their expertise affected patients with DTC-GT, 92% of GCs who felt that their expertise was beneficial gave the following reasons: the limitations of DTC-GT were better understood with genetic counselling (75%) and that it assisted in the clarification of the medical significance of patients’ results (57%) [30]. GCs (6%) agreed or strongly agreed that DTC-GT might threaten the genetic counselling profession, while 79% disagreed or strongly disagreed with this statement [30].
Ethical issues identified by HCPs surrounding DTC-GT
Ethical concerns were identified in 24 of the 27 papers. Ten ethical issues were identified in this review, which overlap with previous HCP perceptions. Of the ten ethical issues, eight were identified in papers from the previous review. The ethical issues found in each paper are summarised in Appendix C. The ethical issues on DTC-GT identified in this review (and a summary of points relating to each issue) are included in Table 3.
The five most identified ethical issues pertaining to DTC-GT were found in at least 10 of the 27 papers identified. These were: clinical utility; clinical validity/reliability; genetic counselling/GP involvement; resource use/resulting downstream costs; misinformation/understanding DTC-GT results. The psychological effects on patients and their behaviours and/or anxieties due to DTC-GT were identified in seven of the papers, while DTC-GT companies’ financial gain/advertising were only mentioned in five of the papers. The option of limiting genetic testing to a clinical setting was mentioned in two papers. Two ethical issues were only reported once each, both in the newly identified papers: 1) DTC-GT threatening the genetic counselling profession and 2) the impact of DTC-GT on the doctor–patient relationship.
Legal issues identified by HCPs surrounding DTC-GT
Legal issues were identified in 12 of the 27 papers, and were divided into four main categories. Potential discrimination on consumers’ employment, health and/or life insurance was mentioned in seven of the 27 papers. Regulation or oversight of regulation of DTC-GT was mentioned in five papers and confidentiality/privacy of genetic information was mentioned in four papers. Lastly, HCPs’ medical and legal responsibilities were mentioned in two of the new papers identified in the updated search. The legal issues found in each paper are summarised in Appendix B. The four legal issues on DTC-GT identified in the included papers (with some explanation for each issue) are included in Table 4.
link